|
The Pfizer/BioNTech and AstraZeneca/Oxford vaccines were the most widely used vaccines to protect the UK population in 2021. Since then most booster doses have been the Pfizer/BioNTech and Moderna vaccines. The AstraZeneca/Oxford, Johnson & Johnson and Valneva vaccines are not currently used.
In total, the World Health Organization (WHO) has approved nine different COVID-19 vaccines for use in a public health emergency. This includes the six approved in the UK, along with three more produced by companies in China and India. These met WHO regulatory standards before being added to their emergency list. There are currently around 380 different vaccines at some stage of the research, development, and testing process.
While the existing vaccines protect people against getting seriously ill, they do not reduce the transmission of emerging new variants virus. And with the virus still circulating, there is an ongoing risk of new variants emerging for which existing vaccines will only provide limited protection against infection.
However, it is highly unlikely that the wall of immunity across the population against severe disease will get significantly less.
Developing a vaccine for COVID-19
Even though COVID-19 is a new disease, caused by a novel coronavirus, work to develop vaccines to protect us against this family of coronaviruses began about 20 years ago. Coronaviruses were already a threat to our health.
The clinical trials to test the vaccines were carried out more quickly than normal by overlapping the different trial stages. A trial’s three stages are usually done one after the other.
No stage was missed out in the COVID vaccine trials and the trials were subject to the same strict regulatory requirements as any other vaccine. This video shows how scientists were able to make the AstraZeneca/Oxford vaccine so quickly.
https://www.youtube.com/embed/ddDiyIKUP0M?wmode=opaque&controls=&rel=0
The vaccines that have been approved for use in the UK were tested in trials with over 20,000 people. In many cases, these trials are larger than trials for other drugs and vaccines which have been licensed.
Please see below for information about how vaccines are developed and how some of the administrative processes were speeded up. With thanks to Nature for permission to use this video.
mRNA vaccines - Pfizer-BioNTech and Moderna
These vaccines each contain the genetic code (mRNA) of the spike protein found on the surface of the SARS-CoV-2 virus. Once inside the body, the spike protein is produced, causing the immune system to recognise it and initiate an immune response.
This means that if the body later encounters the spike protein of the coronavirus, the immune system will recognise it and destroy it before causing infection.
As there is no whole or live virus involved, the vaccines cannot cause COVID-19 disease. The mRNA is naturally degraded after a few days.
The safety and efficacy of the Pfizer-BioNTech vaccine were assessed in clinical trials of over 44,000 people in six countries: USA, Germany, Brazil, Argentina, South Africa and Turkey. The trial also showed that the vaccine provides similar protection in people of all ages, races and ethnicities.
The safety and efficacy of the Moderna vaccine was tested in over 30,000 people across the United States, including older people, people from ethnic minorities and those with underlying health conditions.
Viral vector vaccines - Oxford-AstraZeneca ChAdOx1 nCoV-19 and Janssen
Viral vector vaccines work by delivering the genetic code of the SARS-CoV-2 spike protein to the body’s cells, similar to the mRNA vaccines.
Once inside the body, the spike protein is produced, causing the immune system to recognise it and initiate an immune response. This means that if the body later encounters the spike protein of the coronavirus, the immune system will recognise it and destroy it before causing infection.
This Oxford-AstraZeneca vaccine uses the ChAdOx1 technology, which has been developed and optimised by the Jenner Institute over the last 10 years. This type of vaccine technology has been tested for many other diseases such as influenza (flu), meningococcus, plague and Middle East Respiratory Syndrome (MERS), another type of coronavirus.
The ChAdOx1 nCoV-19 vaccine was tested by the University of Oxford in clinical trials of over 23,000 people in the UK, Brazil and South Africa. A further trial with 40,000 people was also run by AstraZeneca in the USA, Argentina, Chile, Colombia and Peru. Trials were also conducted in India and Japan.
Newer vaccines, such as mRNA vaccines and viral vector vaccines, differ from many traditional vaccines in the way they activate the immune system. Most traditional vaccines inject the antigen (part of the disease that stimulates an immune response) directly into the body.
In contrast, these two newer approaches deliver the genetic instructions for the antigen to the body’s cells. The cells then manufacture the antigen, which goes on to stimulate the immune response.
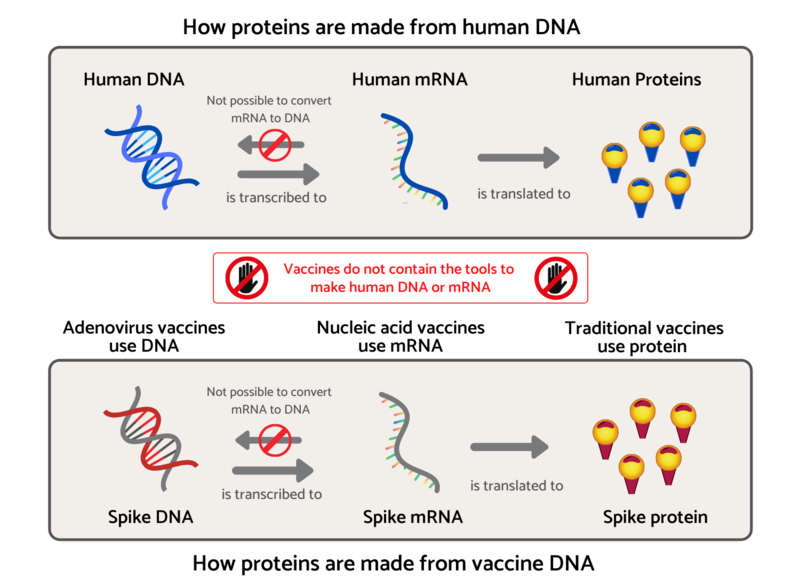
Injecting genetic material has raised questions about the use of these vaccines, such as whether they modify the DNA of those receiving them. However, this is not possible because the code cannot be incorporated into the body’s DNA.
Click here for an accessible text version of this infographic
Protein subunit vaccines – Novavax
Subunit vaccines have been in use for many years, for example against hepatitis B and pertussis (whooping cough). This COVID-19 vaccine contains particles of the spike protein from the virus, along with an adjuvant to help generate a stronger immune response.
Together, these help the body’s immune system to respond to the vaccine and the virus when it encounters it in future.
|